Amarin released detailed REDUCE-IT data this past Saturday and results were mixed. Although the Vascepa active arm showed strong data, the placebo arm used mineral oil, which may have caused problems for patients in control arm. This brings up the question: is the placebo arm making Vascepa data look better than it really is?
Vascepa Active Arm Data Was Solid
Vascepa cut the risk of cardiovascular events (death, heart attack and stroke) by 26%. The death rate from CV causes was reduced by 20%, heart attacks reduced by 31%, strokes by 28%. As seen below, all the endpoints favored Vascepa’s arm, and by a statistically significant measure.
But Questions Surround Placebo
However, the company’s use of mineral oil as their placebo capsule resulted in higher blood fat markers from baseline. Placebo arm showed that:
- LDL (“bad cholesterol) levels rose by 10%
- C-reactive protein (hsCRP), a measure used to calculate heart risk, rose by 30%
- Median APO-B levels, a predictor of CV risk, rose by 8%

This raised many concerns because blood test results on the placebo arm are not supposed to rise, especially when considering that these patients were all on statin therapy. Statins have shown to lowerLDL & apo-b, yet in REDUCE-IT placebo they increased. Now, people doubt whether the mineral oilplacebo might be interfering with the background statin therapy.
To compare, GSK’s Lovaza used olive oil as a placebo in their ASCEND trial (which did not show a statistical significant reduction in CV events). AstraZeneca is using corn oil in their CV event study STRENGTH, with data expected Q4 2019.
It's also important to note that there were no differences in primary or secondary endpoints in the placebo arm for patients with an increase in LDL-C (figure below). Therefore, placebo patients with LDL increase actually had less CV events than those with no change (or a decrease in LDL). This may provide some clarity to support Vascepa's results and ease concerns around the placebo effect.
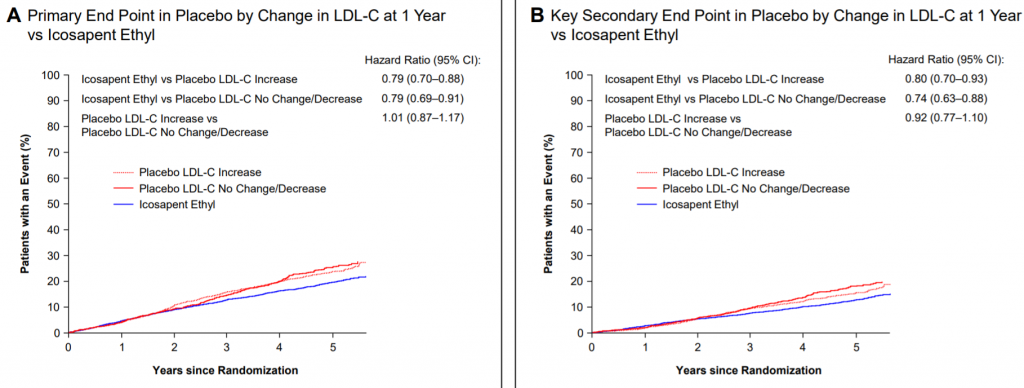
Bottom Line: Although AMRN data was filled with controversy, Vascepa results are still a breakthrough in the CV space. The relatively small difference in LDL between Vascepa group (3% rise in LDL) and placebo (10% rise in LDL) does not fully explain the significant 25% reduction in CV risks that Vascepa provides. Amarin will be a hot take out target with a multi-billion dollar drug.
What Data Means For Acasti?
We believe the questions surrounding Amarin benefit Acasti. Acasti krill oil has shown early promise (read more here) and the company is still about 7 years away from CV outcome trial, if they decide to take that path.
By this time next year, Amarin (mineral oil placebo) and AstraZeneca (corn oil placebo) will have CV outcome data. Acasti can analyze the public data and choose the best placebo to move forward with in their potential CV outcome study.
Additionally, we believe Amarin data continues to validate fish oils (especially EPA ingredient) against CV risks. Acasti has EPA and other lipid lowering ingredients that can yield similar results. ACST will be a name to closely monitor as data nears 2H 2019.
PropThink contributors have no positions in any of the mentioned companies.
Access This Content Now
Sign Up Now!




